DUSP6 (dual specificity phosphatase 6)
2011-09-01 Zhenfeng Zhang , Balazs Halmos AffiliationDivision of Hematology\\\/Oncology, Herbert Irving Comprehensive Cancer Center, New York Presbyterian Hospital-Columbia University Medical Center, New York, NY, USA
Identity
HGNC
LOCATION
12q21.33
LOCUSID
ALIAS
HH19,MKP3,PYST1
FUSION GENES
DNA/RNA
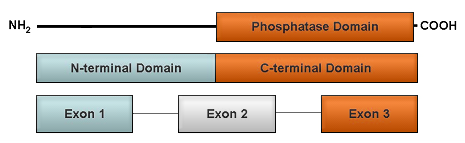
The diagram depicts the structure of the DUSP6 gene (bottom) roughly aligned with its corresponding functional protein domains (middle and top). DUSP6 comprises a C-terminal catalytic domain and an N-terminal non-catalytic domain (middle). The 3 exons of DUSP6 (rectangles) are connected with lines representing introns.
Description
The human DUSP6 gene is located on chromosome 12q21.33 and consists of 3 exons. The full-length coding sequence of DUSP6 contains 1146 nucleotides. The functional phosphatase domain of DUSP6 is encoded by half of exon 2 and almost the entire sequence of exon 3.
Transcription
DUSP6 gene transcription can start from either the first ATG or alternatively the second ATG (Met14), and therefore two protein products are generated which usually demonstrate a double-band appearance in regular immunoblotting assays (Dowd et al., 1998; Zhang et al., 2010).
Proteins

The diagram depicts the structural features of DUSP6. The highly conserved C-terminal domain of DUSP6 contains the canonical tyrosine/threonine-specific phosphatase signature sequence HCXXXXXR at the active site, where the cysteine acts as the essential enzymatic nucleophile and arginine interacts directly with the phosphate group on phosphotyrosine or phosphothreonine (Farooq et al., 2001). The amino-terminal domain of DUSP6 contains a specific arginine-rich kinase interaction motif (KIM) (Tárrega et al., 2005) and a leucine-rich nuclear export signal (NES) necessary and sufficient for nuclear export of the phosphatase (Karlsson et al., 2004).
Description
The full-length DUSP6 protein contains 381 amino acids and has a molecular weight of 44 kDa. DUSPs are characterized by a common structure comprising a C-terminal phosphatase domain that are defined by the active-site signature motif HCXXXXXR. The structure of DUSP proteins confers phosphatase activity for both phosphoserine/threonine and phosphotyrosine residues. An enzyme-dead DUSP6 expression construct can be generated via a 293 Cysteine to Serine/Glycine (C293S/G) point mutation (Wishart et al., 1995; Zhang et al., 2010; Zhou et al., 2006).
Expression
DUSP6 is expressed usually at low level in resting, nonstimulated cells in a variety of tissues and is induced as an early response gene after activation of the ERK-MAPK signaling pathway.
Localisation
DUSP6 is a cytoplasmic dual specificity protein phosphatase.
Function
Mitogen-activated protein kinases (MAPK) constitute a highly conserved family of kinases that relay information from extracellular signals to downstream effectors that control diverse cellular processes such as proliferation, differentiation, migration, survival and apoptosis (Wada and Penninger, 2004). A balance between the activities of upstream activators and various negative regulatory mechanisms of MAPK signaling, which terminate its activation, determines its biological outcomes. DUSP6 is a prototypical member of a subfamily of cytoplasmic MKPs, which includes DUSP7 and DUSP9 as well. These enzymes all display a high degree of substrate selectivity for ERK1 and ERK2 (Keyse, 2008). DUSP6 has been shown to act as a central feedback regulator attenuating ERK levels in developmental programs (Echevarria et al., 2005; Li et al., 2007). The cytoplasmic localization of DUSP6 is mediated by a chromosome region maintenance-1-dependent nuclear export pathway. DUSP6 appears to play a role in determining the subcellular localization of ERK by serving as a cytoplasmic anchor for ERK, thereby mediating a spatio-temporal mechanism of ERK signaling regulation. Cytoplasmic retention of ERK requires both a functional kinase interaction motif and nuclear export site. Defects of these feedback regulation steps are thought to contribute to ERK-MAPK related oncogenesis. An in vivo study has identified DUSP6 as a negative feedback regulator of fibroblast growth factor-stimulated ERK signaling during murine development (Li et al., 2007). Several in vitro studies have demonstrated that DUSP6 acts as a negative regulator of fibroblast growth factor receptor signaling and endothelial cell platelet-derived growth factor receptor signaling via termination of ERK activation (Ekerot et al., 2008; Jurek et al., 2009).
Homology
DUSP6 belongs to a subfamily of ten more closely related dual-specificity MAPK phosphatases (MKPs) within the larger cysteine-dependent dual specificity phosphatase (DUSP) family (Keyse, 2008). While DUSP1 (MKP-1), DUSP4 (MKP-2), and DUSP9 (MKP4) dephosphorylate both ERKs, p38 and JNK, the phosphatases DUSP5 (Hvh-3), DUSP6 (MKP-3), and DUSP7 (MKP-X) exclusively target ERK1/2 MAPKs (Keyse, 2008). The N-terminal domain of all DUSPs has two regions of homology with the Cdc25 cell cycle regulatory phosphatase. The more conserved catalytic domain within DUSPs contains an active site sequence related to the prototypic VH-1 phosphatase encoded by the vaccinia virus. Specificity of MKPs toward MAPKs relies on the KIM domain. Although each MKP targets different subsets of MAPKs, there is an overlap between their specificities (Bermudez et al., 2010).
Mutations
Note
Although DUSP6 has been implicated as a candidate tumor suppressor in several cancer setting, no mutations in the gene have been identified so far.
Implicated in
Entity name
Various cancers
Note
DUSP6 null mice demonstrate enhanced ERK1/2 phosphorylation leading to increased myocyte proliferation and cardiac hypercellularity (Maillet et al., 2008). DUSP6 has been identified as a potential novel tumor suppressor gene in pancreatic cancer since loss of DUSP6 expression might synergize with activating-mutated k-Ras resulting in increased activation of ERK1/2 MAP kinase and thus contribute to the development of the malignant and invasive phenotype in pancreatic cancer (Furukawa et al., 2003). Loss of DUSP6 expression caused by oxidative stress-mediated degradation was also noted in ovarian cancer and correlated with high ERK1/2 activity (Chan et al., 2008). DUSP6 has also been identified as one of only three genes which are uniquely expressed in myeloma cells harboring a constitutively active mutant N-ras gene and is also overexpressed in human melanoma cell lines with potent activating mutations in B-raf and in breast epithelial cells stably expressing H-Ras (Bloethner et al., 2005; Croonquist et al., 2003; Warmka et al., 2004), suggesting that the over-expression of DUSP6 seen in response to activating-mutated Ras or Raf might represent a compensatory increase in the negative feedback control of the ERK1/2 MAPK pathway, which lies downstream of these activated oncogenes. In support of this, the tetracycline-induced expression of a functional fusion protein between DUSP6 and green fluorescent protein in H-ras transformed fibroblasts following injection into nude mice resulted in a large delay in tumor emergence and growth as compared to the untreated control group (Marchetti et al., 2004 ). DUSP6 has been reported to be one of the most highly regulated genes in chronic myeloid leukemia cells upon imatinib treatment (Hakansson et al., 2008) and similarly DUSP6 is overexpressed upon inducible expression of the EGFRvIII oncogene in glioblastoma cells (Ramnarain et al., 2006). DUSP6 has also been demonstrated to be positively correlated with the activity of the oncogenic ERK pathway in non-small cell lung cancer tissue and is an ETS-regulated negative feedback mediator of ERK signaling in lung cancer cells (Zhang et al., 2010).
Prognosis
Elevated DUSP6 RNA expression was reported to be a major negative predictor of survival in patients with resected non-small cell lung cancer as part of a five-gene signature model (Chen et al., 2007).
Article Bibliography
| Pubmed ID | Last Year | Title | Authors |
|---|---|---|---|
| 15186772 | 2004 | Protein tyrosine phosphatases in the human genome. | Alonso A et al |
| 20463170 | 2010 | The dual-specificity MAP kinase phosphatases: critical roles in development and cancer. | Bermudez O et al |
| 15760917 | 2005 | Effect of common B-RAF and N-RAS mutations on global gene expression in melanoma cell lines. | Bloethner S et al |
| 18632752 | 2008 | Loss of MKP3 mediated by oxidative stress enhances tumorigenicity and chemoresistance of ovarian cancer cells. | Chan DW et al |
| 17202451 | 2007 | A five-gene signature and clinical outcome in non-small-cell lung cancer. | Chen HY et al |
| 12791645 | 2003 | Gene profiling of a myeloma cell line reveals similarities and unique signatures among IL-6 response, N-ras-activating mutations, and coculture with bone marrow stromal cells. | Croonquist PA et al |
| 9788880 | 1998 | Isolation of the human genes encoding the pyst1 and Pyst2 phosphatases: characterisation of Pyst2 as a cytosolic dual-specificity MAP kinase phosphatase and its catalytic activation by both MAP and SAP kinases. | Dowd S et al |
| 15572144 | 2005 | Mkp3 is a negative feedback modulator of Fgf8 signaling in the mammalian isthmic organizer. | Echevarria D et al |
| 18321244 | 2008 | Negative-feedback regulation of FGF signalling by DUSP6/MKP-3 is driven by ERK1/2 and mediated by Ets factor binding to a conserved site within the DUSP6/MKP-3 gene promoter. | Ekerot M et al |
| 11239467 | 2001 | Solution structure of ERK2 binding domain of MAPK phosphatase MKP-3: structural insights into MKP-3 activation by ERK2. | Farooq A et al |
| 12759238 | 2003 | Potential tumor suppressive pathway involving DUSP6/MKP-3 in pancreatic cancer. | Furukawa T et al |
| 18181176 | 2008 | Gene expression analysis of BCR/ABL1-dependent transcriptional response reveals enrichment for genes involved in negative feedback regulation. | Håkansson P et al |
| 19106095 | 2009 | Negative and positive regulation of MAPK phosphatase 3 controls platelet-derived growth factor-induced Erk activation. | Jurek A et al |
| 15269220 | 2004 | Both nuclear-cytoplasmic shuttling of the dual specificity phosphatase MKP-3 and its ability to anchor MAP kinase in the cytoplasm are mediated by a conserved nuclear export signal. | Karlsson M et al |
| 18330678 | 2008 | Dual-specificity MAP kinase phosphatases (MKPs) and cancer. | Keyse SM et al |
| 17164422 | 2007 | Dusp6 (Mkp3) is a negative feedback regulator of FGF-stimulated ERK signaling during mouse development. | Li C et al |
| 18753132 | 2008 | DUSP6 (MKP3) null mice show enhanced ERK1/2 phosphorylation at baseline and increased myocyte proliferation in the heart affecting disease susceptibility. | Maillet M et al |
| 15095291 | 2004 | Inducible expression of a MAP kinase phosphatase-3-GFP chimera specifically blunts fibroblast growth and ras-dependent tumor formation in nude mice. | Marchetti S et al |
| 16424019 | 2006 | Differential gene expression analysis reveals generation of an autocrine loop by a mutant epidermal growth factor receptor in glioma cells. | Ramnarain DB et al |
| 16148006 | 2005 | ERK2 shows a restrictive and locally selective mechanism of recognition by its tyrosine phosphatase inactivators not shared by its activator MEK1. | Tárrega C et al |
| 15077147 | 2004 | Mitogen-activated protein kinases in apoptosis regulation. | Wada T et al |
| 15159408 | 2004 | Mitogen-activated protein kinase phosphatase-3 is a tumor promoter target in initiated cells that express oncogenic Ras. | Warmka JK et al |
| 7592916 | 1995 | A single mutation converts a novel phosphotyrosine binding domain into a dual-specificity phosphatase. | Wishart MJ et al |
| 20097731 | 2010 | Dual specificity phosphatase 6 (DUSP6) is an ETS-regulated negative feedback mediator of oncogenic ERK signaling in lung cancer cells. | Zhang Z et al |
| 17046812 | 2006 | Mapping ERK2-MKP3 binding interfaces by hydrogen/deuterium exchange mass spectrometry. | Zhou B et al |
Other Information
Locus ID:
NCBI: 1848
MIM: 602748
HGNC: 3072
Ensembl: ENSG00000139318
Variants:
dbSNP: 1848
ClinVar: 1848
TCGA: ENSG00000139318
COSMIC: DUSP6
RNA/Proteins
Expression (GTEx)
Pathways
Protein levels (Protein atlas)
References
| Pubmed ID | Year | Title | Citations |
|---|---|---|---|
| 38678784 | 2024 | Long non-coding RNA NORAD regulates megakaryocyte differentiation and proplatelet formation via the DUSP6/ERK signaling pathway. | 0 |
| 38821599 | 2024 | Functional Role of DUSP6 in the EGFR-mediated Progression of Head and Neck Squamous Cell Carcinoma. | 0 |
| 38678784 | 2024 | Long non-coding RNA NORAD regulates megakaryocyte differentiation and proplatelet formation via the DUSP6/ERK signaling pathway. | 0 |
| 38821599 | 2024 | Functional Role of DUSP6 in the EGFR-mediated Progression of Head and Neck Squamous Cell Carcinoma. | 0 |
| 36806557 | 2023 | N6-methyadenosine modified SUV39H2 regulates homologous recombination through epigenetic repression of DUSP6 in gastric cancer. | 1 |
| 37029196 | 2023 | DUSP6 is a memory retention feedback regulator of ERK signaling for cellular resilience of human pluripotent stem cells in response to dissociation. | 1 |
| 36806557 | 2023 | N6-methyadenosine modified SUV39H2 regulates homologous recombination through epigenetic repression of DUSP6 in gastric cancer. | 1 |
| 37029196 | 2023 | DUSP6 is a memory retention feedback regulator of ERK signaling for cellular resilience of human pluripotent stem cells in response to dissociation. | 1 |
| 35188450 | 2022 | microRNA-211-5p predicts the progression of postmenopausal osteoporosis and attenuates osteogenesis by targeting dual specific phosphatase 6. | 10 |
| 35189148 | 2022 | DUSP4 Inactivation Leads to Reduced Extracellular Signal‒Regulated Kinase Activity through Upregulation of DUSP6 in Melanoma Cells. | 1 |
| 36462498 | 2022 | SKA1 promotes tumor metastasis via SAFB-mediated transcription repression of DUSP6 in clear cell renal cell carcinoma. | 0 |
| 35188450 | 2022 | microRNA-211-5p predicts the progression of postmenopausal osteoporosis and attenuates osteogenesis by targeting dual specific phosphatase 6. | 10 |
| 35189148 | 2022 | DUSP4 Inactivation Leads to Reduced Extracellular Signal‒Regulated Kinase Activity through Upregulation of DUSP6 in Melanoma Cells. | 1 |
| 36462498 | 2022 | SKA1 promotes tumor metastasis via SAFB-mediated transcription repression of DUSP6 in clear cell renal cell carcinoma. | 0 |
| 32389901 | 2021 | Prevalence and associated phenotypes of DUSP6, IL17RD and SPRY4 variants in a large Chinese cohort with isolated hypogonadotropic hypogonadism. | 5 |
Citation
Zhenfeng Zhang ; Balazs Halmos
DUSP6 (dual specificity phosphatase 6)
Atlas Genet Cytogenet Oncol Haematol. 2011-09-01
Online version: http://atlasgeneticsoncology.org/gene/46105/dusp6
