NUDT6 (nudix (nucleoside diphosphate linked moiety X)-type motif 6)
2010-04-01 Leigh-Ann MacFarlane , Paul Murphy AffiliationDalhousie University, Department of Physiology, Biophysics, Faculty of Medicine, 5850 College Street Sir Charles Tupper Medical Building, Halifax, Nova Scotia B3H 1X5, Canada
DNA/RNA
Note
Human NUDT6 is located on chromosome 4 in the region of q28 on the reverse strand, opposite to FGF-2 gene locus. FGF-2 and NUDT6 genes overlap at 3 ends, and the mRNAs form a sense-antisense pair. The NUDT6 mRNA (referred to as FGF-AS) has been implicated in the regulation of FGF2 mRNA stability.
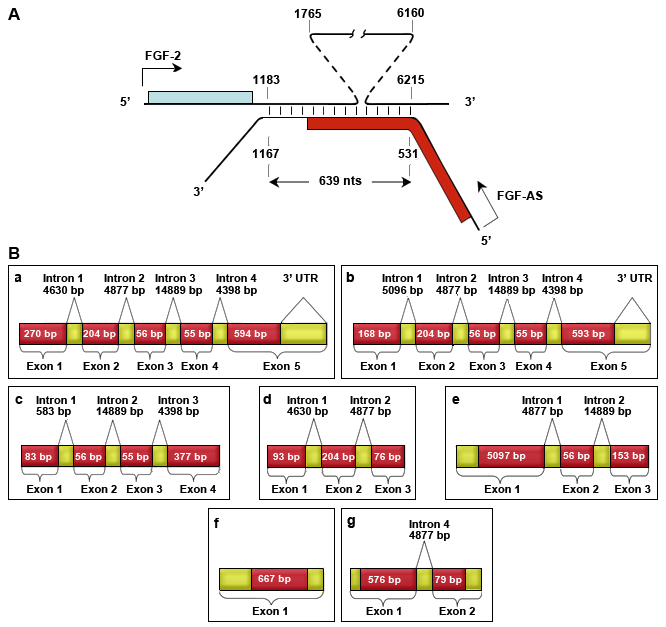
Figure A. The schematic representation of the overlap between human NUDT6 (FGF-AS) and FGF2 gene transcripts (colored boxes, coding region; connecting vertical lines, complementary regions between transcripts). Adapted from: MacFarlane LA, et al., 2010. Molecular Endocrinology 24.
Figure B. The schematic representation of the human NUDT6 gene transcripts, variants a-g (red boxes, coding region; yellow boxes, untranslated region).
Description
The human NUDT6 gene is 34271 bp in length, composed of a 5UTR, 16 exons, 6 introns and a 3UTR. The 5 and 3UTR contain a variety of regulatory elements that regulate NUDT6 expression. NUDT6 gene transcription is regulated by a core promoter mapping from -1871 to +181 (relative to the transcription start site +1, up-stream -), which is 44 kb downstream from the FGF-2 promoter, however the proximal -151/+181 region confers almost full transcription activity.
The promoter lacks a consensus TATA box or CCAAT element. The region between the first two exons contains two Sp1 transcription factor binding sites (-372/-58 relative to the first exon start site). The common upstream region (all subsequent positions relative to first exon start site) from these start sites contains a multitude of tissue specific transcription factor binding sites, which include lymphocyte specific factors Ets at -229 and -83, GATA at -662 and +56, Lyf-1 at -981; skeletal muscle consensus E-boxes at -901 and +30; cardiac factor Nkx-2.5 at -1501 and -582; liver and adipose C/EBP factor at -624; and testis specific factors SRY at -1740, -671, +163, +171 and Sox-5 at -1472, -632.
Two negative regulatory elements also reside in this shared upstream region, at -1871 and -1315. The NUDT6 3UTR contains a singe AU-rich element (ARE) and seven AU-rich-like sequences which negatively regulate mRNA stability. A portion of the NUDT6 coding region and 3UTR (+531/+1167) is fully complementary to the 3UTR of FGF2 and interaction through this region leads to the formation of a sense-antisense pair.
The promoter lacks a consensus TATA box or CCAAT element. The region between the first two exons contains two Sp1 transcription factor binding sites (-372/-58 relative to the first exon start site). The common upstream region (all subsequent positions relative to first exon start site) from these start sites contains a multitude of tissue specific transcription factor binding sites, which include lymphocyte specific factors Ets at -229 and -83, GATA at -662 and +56, Lyf-1 at -981; skeletal muscle consensus E-boxes at -901 and +30; cardiac factor Nkx-2.5 at -1501 and -582; liver and adipose C/EBP factor at -624; and testis specific factors SRY at -1740, -671, +163, +171 and Sox-5 at -1472, -632.
Two negative regulatory elements also reside in this shared upstream region, at -1871 and -1315. The NUDT6 3UTR contains a singe AU-rich element (ARE) and seven AU-rich-like sequences which negatively regulate mRNA stability. A portion of the NUDT6 coding region and 3UTR (+531/+1167) is fully complementary to the 3UTR of FGF2 and interaction through this region leads to the formation of a sense-antisense pair.
Transcription
The primary transcript can be alternatively spliced to produce at least 7 splice variants, a-d. Full length variants a and b only differ in the use of an alternative first exon, designated 1A or 1B. The 3 ends of the variants share sequence similarity. Two transcriptional start sites have been identified, one 15 bp upstream of the 1A exon (designated +1) and the other 84 bp upstream of the 1B exon (+312). It is unclear whether another transcription start site specific for other variants are located further downstream.
NUDT6 transcripts are often designated FGF-AS (FGF antisense). The two longest transcripts, a and b, are classified as cis-antisense because they are transcribed from the same gene locus, on the opposite DNA strand and their 3UTR is fully complementary to the 3UTR of FGF-2 over two regions 583 bp and 56 bp in length.
NUDT6 transcripts are often designated FGF-AS (FGF antisense). The two longest transcripts, a and b, are classified as cis-antisense because they are transcribed from the same gene locus, on the opposite DNA strand and their 3UTR is fully complementary to the 3UTR of FGF-2 over two regions 583 bp and 56 bp in length.
Pseudogene
NA.
Proteins
Note
Human NUDT6 encodes 3 novel nudix proteins with unknown function.

Schematic representing predicted NUDT6 isoforms encoded by alternate splice RNA transcripts (deep blue boxes, nudix motif; light blue box, MTSP-mitochondrial targeting signal peptide).
Description
NUDT6 splice variants a-c contain open reading frames (ORF) that predict isoforms of a novel nudix motif protein, originally designated GFG. The nudix box motif is defined by the consensus signature amino acid sequence GX5EX7REUXEEXGU, where X is any amino acid and U is a bulky hydrophobic amino acid, usually isoleucine, leucine or valine. To date, three different molecular weight isoforms have been identified in human, of 35, 28 and 17 kDa, which are presumably generated by alternative translation initiation. Isoforms are designated as a, b or c however, this does not necessarily indicate that the isoform was synthesized from the corresponding transcript variant. The 35 kDa isoform is synthesized from the full length FGF-ASa, by translation initiation from the in-frame AUG codon located in exon 1A. The origin of the 28 kDa isoform is unclear. It is suspected that it is synthesized from an in-frame CUG codon in exon 2 of either FGF-AS a or b.
However, it is possible that the 28 kDa product is a proteolytic fragment of the 35 kDa isoform. The 17 kDa isoform may arise from translation initiation at an in-frame CUG codon in exon 3 of FGF-ASb or AUG codon in the first exon of FGF-ASc. The NUDT6 isoforms are detected as stable homo- and hetero-dimers by western blotting, which can be disrupted by dithiothreitol (DTT) and boiling. Potential dimerization domains have been mapped to both the N-terminus and COOH-terminus of NUDT6.
However, it is possible that the 28 kDa product is a proteolytic fragment of the 35 kDa isoform. The 17 kDa isoform may arise from translation initiation at an in-frame CUG codon in exon 3 of FGF-ASb or AUG codon in the first exon of FGF-ASc. The NUDT6 isoforms are detected as stable homo- and hetero-dimers by western blotting, which can be disrupted by dithiothreitol (DTT) and boiling. Potential dimerization domains have been mapped to both the N-terminus and COOH-terminus of NUDT6.
Expression
NUDT6 is expressed in a tissue and developmental stage specific manner. RNA transcripts are detected in most human tissues including liver, thymus, spleen, peripheral blood leukocytes, heart skeletal muscle, testis, colon and kidney. However, which transcript variants are expressed appears to be tissue specific. The full length FGF-ASb is thought to be the predominant variant in most tissues however variant FGF-ASa is the major variant in normal hematopoietic tissues. Furthermore, some tissues co-express FGF-2 and the ratio between FGF-2 and FGF-AS transcripts varies with tissue and development stage. FGF-AS levels are relatively low in many embryonic tissues, with expression increasing dramatically in a tissue specific manner postnatally. FGF-2 and FGF-AS exhibit an inverse relationship in normal tissues, tumor cell lines, embryonic development and throughout cell cycle progression.
The level of NUDT6 expression and its ratio with FGF-2 expression is frequently altered in tumors. Normal pituitary expresses moderate levels of NUDT6 and no FGF-2 while pituitary tumors have reduced NUDT6 expression and high levels of FGF-2. The NUDT6/FGF-2 expression ratio decreases dramatically in tumors compared to normal tissue. Varying NUDT6/FGF-2 ratios have also been observed in esophageal adenocarcinomas. Additionally, transient increase in NUDT6 expression occurs in response to treatment with interleukin-2 and prolactin.
The level of NUDT6 expression and its ratio with FGF-2 expression is frequently altered in tumors. Normal pituitary expresses moderate levels of NUDT6 and no FGF-2 while pituitary tumors have reduced NUDT6 expression and high levels of FGF-2. The NUDT6/FGF-2 expression ratio decreases dramatically in tumors compared to normal tissue. Varying NUDT6/FGF-2 ratios have also been observed in esophageal adenocarcinomas. Additionally, transient increase in NUDT6 expression occurs in response to treatment with interleukin-2 and prolactin.
Localisation
NUDT6 can reside in the mitochondria, cytoplasm and nucleus, however its subcellular localization varies with isoform, cell type, disease state and extracellular stimulus. NUDT6a predominantly localizes to mitochondria whereas NUDT6b and NUDT6c primarily reside in the cytoplasm and nucleus. NUDT6 is only found in the cytoplasm of normal esophageal squamous epithelial cells whereas in normal lymphocytes it is exclusively nuclear. However, transformation of these cells results in the redistribution of NUDT6. Cells from esophageal adenocarcinoma tumors and lymph nodes of patient with immunoblastic lymphoma localize NUDT6 to the nucleus and cytoplasm.
Function
The RNA and protein products appear to have distinct biological functions. NUDT6 mRNA (FGF-AS) plays a role in FGF-2 regulation, proliferation, and cell survival. Additionally, the NUDT6 protein has been implicated in the control of hormone production in the pituitary, and possibly in the removal of potentially hazardous compounds and metabolites by virtue of its conserved nudix domain. However, it is not always clear whether a specific action is a result of the RNA or protein function and this is further complicated by multiple antisense splice variants and protein isoforms.
FGF-AS regulates FGF-2 transcript stability. Although the details of the mechanism involved are unclear, evidence suggest involvement of RNA interference and/or a dsRNA duplex formed between the 3UTRs of FGF-AS and FGF-2. In addition to regulating FGF-2 abundance it has been suggested that it also controls FGF-2 isoform translation and localization. The regulatory role of FGF-AS over FGF-2 is thought to account for observed effects on cell proliferation and survival.
NUDT6 protein is a nudix hydrolase which is a class of "house cleaning" enzymes capable of hydrolyzing a broad range of substrates, all defined as nucleoside diphosphates linked to some other moiety, that include nucleoside di- and triphosphates, dinucleoside and diphosphoinositol polyphosphates, nucleotide sugars and RNA caps. The substrate of human NUDT6 has yet to be elucidated and therefore its physiological function remains unknown. NUDT6 has observed effects on cell proliferation independent of those associated with FGF-AS. NUDT6 overexpression in human colorectal cancer cells increases proliferation. Perhaps NUDT6s effects on proliferation are dependent on expression level, isoform and/or cell type, as is the case with FGF-2. Furthermore, NUDT6 is involved in hormone production. GFG expression can increase levels of prolactin. However it is unclear if these effects are mediated through the same MAPK pathway utilized by FGF-2 to increase prolactin expression.
Additionally, NUDT6 alters the isoform ratio of growth hormone, by increasing synthesis of the 22 kDa isoform and not the 20 kDa.
FGF-AS regulates FGF-2 transcript stability. Although the details of the mechanism involved are unclear, evidence suggest involvement of RNA interference and/or a dsRNA duplex formed between the 3UTRs of FGF-AS and FGF-2. In addition to regulating FGF-2 abundance it has been suggested that it also controls FGF-2 isoform translation and localization. The regulatory role of FGF-AS over FGF-2 is thought to account for observed effects on cell proliferation and survival.
NUDT6 protein is a nudix hydrolase which is a class of "house cleaning" enzymes capable of hydrolyzing a broad range of substrates, all defined as nucleoside diphosphates linked to some other moiety, that include nucleoside di- and triphosphates, dinucleoside and diphosphoinositol polyphosphates, nucleotide sugars and RNA caps. The substrate of human NUDT6 has yet to be elucidated and therefore its physiological function remains unknown. NUDT6 has observed effects on cell proliferation independent of those associated with FGF-AS. NUDT6 overexpression in human colorectal cancer cells increases proliferation. Perhaps NUDT6s effects on proliferation are dependent on expression level, isoform and/or cell type, as is the case with FGF-2. Furthermore, NUDT6 is involved in hormone production. GFG expression can increase levels of prolactin. However it is unclear if these effects are mediated through the same MAPK pathway utilized by FGF-2 to increase prolactin expression.
Additionally, NUDT6 alters the isoform ratio of growth hormone, by increasing synthesis of the 22 kDa isoform and not the 20 kDa.
Homology
NUDT6 contains a conserved nudix motif common to other members of the Nudix family of phosphohydrolases. The Nudix motif is GXXXXXEXXXXXXXREUXEEXGU where U is Isoleucine, Leucine, or Valine and X is any amino acid. NUDT6 is highly conserved among man, cow, mouse, worm, and fruit fly, and GFG homologs across species are more evolutionarily related to each other than to other nudix proteins from the same species.
Implicated in
Entity name
Esophageal adenocarcinoma
Note
Esophageal adenocarcinoma refers to uncontrolled growth of glandular cells in the esophagus and the junction between the esophagus and the stomach.
Prognosis
Elevated expression of FGF-AS in FGF-2 expressing esophageal adenocarcinomas is associated with reduced tumor reoccurrence following surgical resection of tumors and increased survival rates, suggesting that it may be used as a prognostic indicator.
Oncogenesis
Esophageal adendocarcinoma tumors overexpressed FGF-AS and cytoplasmic GFG in comparison to normal match esophageal tissue. However, the reduced tumor reoccurrence and improved survival rates specifically correlated to FGF-AS levels, not GFG levels. Evidence suggests FGF-AS tumor suppressive role is a result of its post-transcription control over FGF-2.
Entity name
Melanoma
Note
Melanoma is a malignant tumor of melanocytes, which are found primarily in the skin, however they can develop in melanocytes found in the eye and bowel. A characteristic of aggressive melanomas is their ability to form fluid-conducting vasculogenic-like networks.
Oncogenesis
A preliminary study investigating these 3D tubular networks within tumors identified NUDT6 as one of the many genes overexpressed in aggressive melanomas and speculated it is involved in promoting self-renewal and tumor cell plasticity in melanoma cancer networks. Additionally, they suggest FGF-AS could play a role in the development of the endothelia-lined vasculature networks in melanomas indirectly through its regulatory control over FGF-2 expression, which is associated with angiogenesis, proliferation and survival.
Entity name
Colorectal cancer
Note
Colorectal cancer refers to uncontrolled growth of cells that line the colon, rectum and appendix, collectively the large intestine.
Oncogenesis
Induced overexpression of NUDT6 in a variety of human colorectal cells significantly increases cancer cell proliferation and their clonogenic capacity. NUDT6 is described as having tumor promoting functions in this cellular environment and it is suggested that it plays a role in colorectal cancer development and progression.
Entity name
Endometriosis
Note
Endometriosis is a medical condition affecting the endometrium lining of the uterus. The endometrium is comprised of hormonally responsive cells that proliferate and secrete under the influence of estrogen and progesterone. Upon menstruation the endometrium lining is shed as a part of the menstrual flow. Endometriosis describes the presence of endometrial cells outside of the uterus, such as the ovaries, fallopian tubes, bladder and interstitial space in the abdominal cavity.
Patients with endometriosis lesions have reduced FGF-AS-b mRNA levels and elevated FGF-2 mRNA levels during the late proliferative phase of the menstrual cycle, compared to control patients. This increased FGF/FGF-AS ratio is thought to contribute to the development of endometriosis.
Patients with endometriosis lesions have reduced FGF-AS-b mRNA levels and elevated FGF-2 mRNA levels during the late proliferative phase of the menstrual cycle, compared to control patients. This increased FGF/FGF-AS ratio is thought to contribute to the development of endometriosis.
Article Bibliography
| Pubmed ID | Last Year | Title | Authors |
|---|---|---|---|
| 11266510 | 2001 | The endogenous fibroblast growth factor-2 antisense gene product regulates pituitary cell growth and hormone production. | Asa SL et al |
| 15725477 | 2005 | Coexpression and regulation of the FGF-2 and FGF antisense genes in leukemic cells. | Baguma-Nibasheka M et al |
| 16278388 | 2005 | Basic fibroblast growth factor (FGF-2) overexpression is a risk factor for esophageal cancer recurrence and reduced survival, which is ameliorated by coexpression of the FGF-2 antisense gene. | Barclay C et al |
| 8810257 | 1996 | The MutT proteins or "Nudix" hydrolases, a family of versatile, widely distributed, "housecleaning" enzymes. | Bessman MJ et al |
| 18655191 | 2008 | Microgenomics profile the endogenous angiogenic phenotype in subpopulations of aggressive melanoma. | Demou ZN et al |
| 10022609 | 1999 | Characterization of the promoter for the human antisense fibroblast growth factor-2 gene; regulation by Ets in Jurkat T cells. | Gagnon ML et al |
| 9246680 | 1997 | Regulation of gene expression by natural antisense RNA transcripts. | Knee R et al |
| 7999082 | 1994 | Basic fibroblast growth factor sense (FGF) and antisense (gfg) RNA transcripts are expressed in unfertilized human oocytes and in differentiated adult tissues. | Knee RS et al |
| 20197313 | 2010 | Regulation of fibroblast growth factor-2 by an endogenous antisense RNA and by argonaute-2. | MacFarlane LA et al |
| 12788899 | 2003 | Different basic fibroblast growth factor and fibroblast growth factor-antisense expression in eutopic endometrial stromal cells derived from women with and without endometriosis. | Mihalich A et al |
| 7984147 | 1994 | Identification and characterization of an antisense RNA transcript (gfg) from the human basic fibroblast growth factor gene. | Murphy PR et al |
| 19157820 | 2010 | A potential proliferative gene, NUDT6, is down-regulated by green tea catechins at the posttranscriptional level. | Sukhthankar M et al |
| 17569023 | 2007 | Alternative splicing of the FGF antisense gene: differential subcellular localization in human tissues and esophageal adenocarcinoma. | Zhang SC et al |
Other Information
Locus ID:
NCBI: 11162
MIM: 606261
HGNC: 8053
Ensembl: ENSG00000170917
Variants:
dbSNP: 11162
ClinVar: 11162
TCGA: ENSG00000170917
COSMIC: NUDT6
RNA/Proteins
Expression (GTEx)
Protein levels (Protein atlas)
References
| Pubmed ID | Year | Title | Citations |
|---|---|---|---|
| 37343711 | 2023 | NUDT6 and NUDT9, two mitochondrial members of the NUDIX family, have distinct hydrolysis activities. | 0 |
| 37343711 | 2023 | NUDT6 and NUDT9, two mitochondrial members of the NUDIX family, have distinct hydrolysis activities. | 0 |
| 20406964 | 2010 | Risk of meningioma and common variation in genes related to innate immunity. | 21 |
| 20438785 | 2010 | Polymorphisms in innate immunity genes and risk of childhood leukemia. | 12 |
| 20628086 | 2010 | Variation at the NFATC2 locus increases the risk of thiazolidinedione-induced edema in the Diabetes REduction Assessment with ramipril and rosiglitazone Medication (DREAM) study. | 15 |
| 20800603 | 2010 | Investigation of genetic susceptibility factors for human longevity - a targeted nonsynonymous SNP study. | 14 |
| 20406964 | 2010 | Risk of meningioma and common variation in genes related to innate immunity. | 21 |
| 20438785 | 2010 | Polymorphisms in innate immunity genes and risk of childhood leukemia. | 12 |
| 20628086 | 2010 | Variation at the NFATC2 locus increases the risk of thiazolidinedione-induced edema in the Diabetes REduction Assessment with ramipril and rosiglitazone Medication (DREAM) study. | 15 |
| 20800603 | 2010 | Investigation of genetic susceptibility factors for human longevity - a targeted nonsynonymous SNP study. | 14 |
| 19054571 | 2009 | Polymorphisms in genes involved in neurodevelopment may be associated with altered brain morphology in schizophrenia: preliminary evidence. | 23 |
| 19416273 | 2009 | Association study between keratinocyte-derived growth factor gene polymorphisms and susceptibility to vitiligo vulgaris in a Taiwanese population: potential involvement of stem cell factor. | 5 |
| 19423540 | 2009 | Common variation in genes related to innate immunity and risk of adult glioma. | 35 |
| 19913121 | 2009 | Gene-centric association signals for lipids and apolipoproteins identified via the HumanCVD BeadChip. | 105 |
| 19054571 | 2009 | Polymorphisms in genes involved in neurodevelopment may be associated with altered brain morphology in schizophrenia: preliminary evidence. | 23 |
Citation
Leigh-Ann MacFarlane ; Paul Murphy
NUDT6 (nudix (nucleoside diphosphate linked moiety X)-type motif 6)
Atlas Genet Cytogenet Oncol Haematol. 2010-04-01
Online version: http://atlasgeneticsoncology.org/gene/41593/nudt6
