TRPV1 (transient receptor potential cation channel, subfamily V, member 1)
2010-11-01 Massimo Nabissi , Giorgio Santoni AffiliationSchool of Pharmacy, Section of Experimental Medicine, University of Camerino, 62032 Camerino (MC), Italy
Identity

DNA/RNA
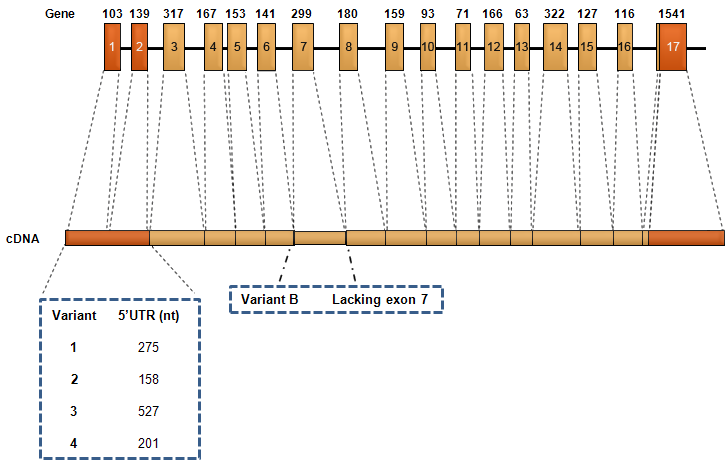
Description
Transcription
Proteins
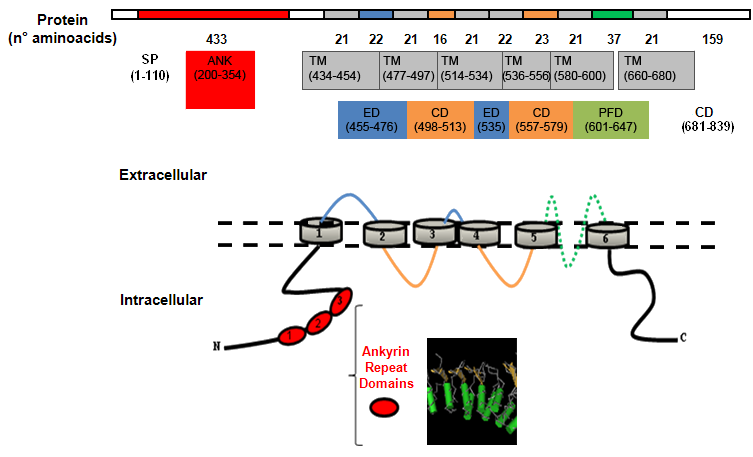
Description
| Aminoacid | Residue modification |
| 117 | Phosphoserine |
| 145 | Phosphoserine |
| 371 | Phosphoserine |
| 502 | Phosphoserine |
| 604 | Glycosylation |
| 705 | Phosphoserine |
| 775 | Phosphoserine |
| 801 | Phosphoserine |
| 821 | Phosphoserine |
Expression
Localisation
Function
TRPV1 antagonists. Natural TRPV1 antagonists are actually restricted to two plant derived compounds, the thapsigargin that is the irritant principle of Thapsia garganica L. and yohimbine, an indole alkaloid from the tree Corynanthe yohimbe K. The endogenous TRPV1 antagonists discovery up to now are dynorphins, adenosine, various dietary omega-3 fatty acids like eicosapentaenoic and linolenic acids, the endogenous fatty acid amide hydrolase (FAAH) and different polyamines as putrescine, spermidine, and spermine permeate. The most active non-natural compound that act as TRPV1 antagonist are capsazepine and 5-iodoRTX.
Ligand-binding site. By comparative analysis of the primary structure of theTRPV1 and by mutagenesis studies has been revealed a critical role for Tyr511 and Ser512 (between the second intracellular loop and TM3), confirming that the vanilloid binding site is located intracellulary, moreover a third critical residue in the putative TM4 segment (Leu547) was indicated as relevant in ligand-binding.
The effect of extracellular protons (as Ca2+), acts primarily by increasing channel opening, rather than interacting directly with the vanilloid binding site.
Related TRPV1 intracellular signaling pathways
EGFR (epidermal growth factor receptor). TRPV1 has been found to down-regulate epidermal growth factor receptor (EGFR) expression. Interaction of TRPV1 terminal cytosolic domain with EGFR induces EGFR ubiquitination and degradation. Moreover, by transfection of TRPV1 in HEK293 cells a decreased EGFR protein expression was observed (Bode et al., 2009).
Fas/CD95. Activation of TRPV1 with capsaicin, in low-grade urothelial cancer cells, induced a TRPV1-dependent G0/G1 cell cycle arrest and apoptosis by inducing transcription of pro-apoptotic genes Fas/CD95, Bcl-2 and caspases, and by activation of the DNA damage response pathway. Moreover, CPS stimulation induced a TRPV1-dependent redistribution and its clustering with Fas/CD95. In addition, an involvement of capsaicin in activation of the ATM kinase/p53 pathways was found (Amantini et al., 2009).
PKA (protein kinase A). TRPV1 are found phosphorylated by PKA in the amino terminus Ser116 and Thr370 and involved in desensitisation while phosphorylation of Ser116 by PKA inhibits dephosphorylation of TRPV1 caused by capsaicin exposure (Mohapatra and Nau, 2003).
PKC (protein kinase C). Several inflammatory mediators, like ATP, bradykinin, prostaglandins and trypsin or tryptase activated Gq coupled receptors and induced PKC-dependent phosphorylation of TRPV1 (Moriyama et al., 2003). PKC dependent phosphorylation of TRPV1 potentiates capsaicin- or proton-evoked responses and reduces temperature threshold for TRPV1 activation. Direct phosphorylation of TRPV1 by PKC has been located at Ser502 and Ser800 (Bhave et al., 2003).
IGF-I (insulin growth factor I). Insulin and IGF-I increase translocation of TRPV1 to the plasma membrane via activation of IGF receptors, which, in turn, induced PI(3) kinase and PKC activation (Van Buren et al., 2005).
CdK5 (cyclin-dependent kinase 5). CdK5 can directly phosphorylate Thr407 in TRPV1, while inhibition of CdK5 activity decreases TRPV1 function and Ca2+ influx (Pareek et al., 2007).
Homology
Implicated in
Article Bibliography
| Pubmed ID | Last Year | Title | Authors |
|---|---|---|---|
| 19502594 | 2009 | Triggering of transient receptor potential vanilloid type 1 (TRPV1) by capsaicin induces Fas/CD95-mediated apoptosis of urothelial cancer cells in an ATM-dependent manner. | Amantini C et al |
| 15708075 | 2005 | Capsaicin receptor TRPV1 in urothelium of neurogenic human bladders and effect of intravesical resiniferatoxin. | Apostolidis A et al |
| 14523239 | 2003 | Protein kinase C phosphorylation sensitizes but does not activate the capsaicin receptor transient receptor potential vanilloid 1 (TRPV1). | Bhave G et al |
| 12142852 | 2002 | Pain perception in patients with schizophrenia. | Blumensohn R et al |
| 19155296 | 2009 | Transient receptor potential type vanilloid 1 suppresses skin carcinogenesis. | Bode AM et al |
| 8108379 | 1993 | Hundreds of ankyrin-like repeats in functionally diverse proteins: mobile modules that cross phyla horizontally? | Bork P et al |
| 11418861 | 2001 | Bradykinin and nerve growth factor release the capsaicin receptor from PtdIns(4,5)P2-mediated inhibition. | Chuang HH et al |
| 15047233 | 2004 | Arachidonyl ethanolamide induces apoptosis of uterine cervix cancer cells via aberrantly expressed vanilloid receptor-1. | Contassot E et al |
| 18830626 | 2009 | Increased expressions of cannabinoid receptor-1 and transient receptor potential vanilloid-1 in human prostate carcinoma. | Czifra G et al |
| 19778904 | 2009 | The endoplasmic reticulum of dorsal root ganglion neurons contains functional TRPV1 channels. | Gallego-Sandín S et al |
| 14996838 | 2004 | Molecular determinants of vanilloid sensitivity in TRPV1. | Gavva NR et al |
| 15788769 | 2005 | Selective blockade of the capsaicin receptor TRPV1 attenuates bone cancer pain. | Ghilardi JR et al |
| 17241282 | 2007 | Capsaicin-sensitive sensory fibers in the islets of Langerhans contribute to defective insulin secretion in Zucker diabetic rat, an animal model for some aspects of human type 2 diabetes. | Gram DX et al |
| 16174661 | 2006 | Vanilloids in pancreatic cancer: potential for chemotherapy and pain management. | Hartel M et al |
| 20932260 | 2011 | TRP channels in the digestive system. | Holzer P et al |
| 16517187 | 2006 | Enhanced formation of nitric oxide in bladder carcinoma in situ and in BCG treated bladder cancer. | Hosseini A et al |
| 19320840 | 2009 | Increased expression of TRPV1 in squamous cell carcinoma of the human tongue. | Marincsák R et al |
| 20021438 | 2008 | TRPV1: on the road to pain relief. | Jara-Oseguera A et al |
| 21083604 | 2010 | Expression of transient receptor potential vanilloid-1 (TRPV1) in urothelial cancers of human bladder: relation to clinicopathological and molecular parameters. | Kalogris C et al |
| 15992990 | 2005 | Transient receptor potential vanilloid type 1 (TRPV1) expression changes from normal urothelium to transitional cell carcinoma of human bladder. | Lazzeri M et al |
| 17582331 | 2007 | The ankyrin repeats of TRPV1 bind multiple ligands and modulate channel sensitivity. | Lishko PV et al |
| 19059400 | 2009 | Capsaicin, a component of red peppers, induces expression of androgen receptor via PI3K and MAPK pathways in prostate LNCaP cells. | Malagarie-Cazenave S et al |
| 18786439 | 2008 | High expression of vanilloid receptor-1 is associated with better prognosis of patients with hepatocellular carcinoma. | Miao X et al |
| 14506258 | 2003 | Desensitization of capsaicin-activated currents in the vanilloid receptor TRPV1 is decreased by the cyclic AMP-dependent protein kinase pathway. | Mohapatra DP et al |
| 12853424 | 2003 | Possible involvement of P2Y2 metabotropic receptors in ATP-induced transient receptor potential vanilloid receptor 1-mediated thermal hypersensitivity. | Moriyama T et al |
| 17656027 | 2007 | Bone cancer increases transient receptor potential vanilloid subfamily 1 expression within distinct subpopulations of dorsal root ganglion neurons. | Niiyama Y et al |
| 17194758 | 2007 | Cyclin-dependent kinase 5 modulates nociceptive signaling through direct phosphorylation of transient receptor potential vanilloid 1. | Pareek TK et al |
| 16596770 | 2005 | Pharmacological actions of cannabinoids. | Pertwee RG et al |
| 15098930 | 2004 | Role of vanilloid receptors in the capsaicin-mediated induction of iNOS in PC12 cells. | Qiao S et al |
| 19914218 | 2010 | Cannabidiol inhibits cancer cell invasion via upregulation of tissue inhibitor of matrix metalloproteinases-1. | Ramer R et al |
| 17174891 | 2006 | TRPV1+ sensory neurons control beta cell stress and islet inflammation in autoimmune diabetes. | Razavi R et al |
| 18297068 | 2008 | A single N-terminal cysteine in TRPV1 determines activation by pungent compounds from onion and garlic. | Salazar H et al |
| 16374544 | 2006 | Induction of apoptosis in prostate tumor PC-3 cells and inhibition of xenograft prostate tumor growth by the vanilloid capsaicin. | Sánchez AM et al |
| 15913603 | 2005 | Expression of the transient receptor potential vanilloid 1 (TRPV1) in LNCaP and PC-3 prostate cancer cells and in human prostate tissue. | Sanchez MG et al |
| 16645193 | 2006 | Transduction mechanisms in airway sensory nerves. | Taylor-Clark T et al |
| 15857517 | 2005 | Sensitization and translocation of TRPV1 by insulin and IGF-I. | Van Buren JJ et al |
| 15128293 | 2004 | Endovanilloids. Putative endogenous ligands of transient receptor potential vanilloid 1 channels. | Van Der Stelt M et al |
| 19305789 | 2008 | Herbal compounds and toxins modulating TRP channels. | Vriens J et al |
| 16836986 | 2006 | Sensory fibers containing vanilloid receptor-1 (VR-1) mediate spinal cord stimulation-induced vasodilation. | Wu M et al |
| 16354198 | 2005 | Noxious heat and scratching decrease histamine-induced itch and skin blood flow. | Yosipovitch G et al |
Other Information
Locus ID:
NCBI: 7442
MIM: 602076
HGNC: 12716
Ensembl: ENSG00000196689
Variants:
dbSNP: 7442
ClinVar: 7442
TCGA: ENSG00000196689
COSMIC: TRPV1
RNA/Proteins
Expression (GTEx)
Pathways
PharmGKB
| Entity ID | Name | Type | Evidence | Association | PK | PD | PMIDs |
|---|---|---|---|---|---|---|---|
| PA164754825 | botulinum toxin type a | Chemical | ClinicalAnnotation | associated | PD | ||
| PA165108779 | Migraine NOS | Disease | ClinicalAnnotation | associated | PD | ||
| PA445208 | Pain | Disease | ClinicalAnnotation | associated | PD | 30908574 | |
| PA448015 | acetaminophen | Chemical | ClinicalAnnotation | associated | PD | 30908574 |
References
| Pubmed ID | Year | Title | Citations |
|---|---|---|---|
| 37902941 | 2024 | TRPV1 inhibition suppresses non-small cell lung cancer progression by inhibiting tumour growth and enhancing the immune response. | 1 |
| 37994506 | 2024 | Transient receptor potential vanilloid 1: A potential therapeutic target for the treatment of osteoarthritis and rheumatoid arthritis. | 4 |
| 38047304 | 2024 | TRPV1 enhances cholecystokinin signaling in primary vagal afferent neurons and mediates the central effects on spontaneous glutamate release in the NTS. | 0 |
| 38234046 | 2024 | LncRNA SNHG1 alleviates myocardial ischaemia-reperfusion injury by regulating the miR-137-3p/KLF4/TRPV1 axis. | 2 |
| 38339203 | 2024 | TRPV1 Channel in Human Eosinophils: Functional Expression and Inflammatory Modulation. | 0 |
| 38389848 | 2024 | 20-HETE mediated TRPV1 activation drives allokinesis via MrgprA3(+) neurons in chronic dermatitis. | 0 |
| 38440794 | 2024 | Relation Between Reactive Oxygen Species Production and Transient Receptor Potential Vanilloid1 Expression in Human Skin During Aging. | 2 |
| 38497183 | 2024 | Low-Level miR-199 Contribute to Neuropathic Low Back Pain via TRPV1 by Regulating the Production of Pro-Inflammatory Cytokines on Macrophage. | 0 |
| 38531262 | 2024 | TRPV1: Receptor structure, activation, modulation and role in neuro-immune interactions and pain. | 2 |
| 38631651 | 2024 | Transient receptor potential vanilloid-1 (TRPV1) channels act as suppressors of the growth of glioma. | 0 |
| 38733310 | 2024 | FGF13 enhances the function of TRPV1 by stabilizing microtubules and regulates acute and chronic itch. | 0 |
| 38927688 | 2024 | Association between Variants of the TRPV1 Gene and Body Composition in Sub-Saharan Africans. | 0 |
| 37902941 | 2024 | TRPV1 inhibition suppresses non-small cell lung cancer progression by inhibiting tumour growth and enhancing the immune response. | 1 |
| 37994506 | 2024 | Transient receptor potential vanilloid 1: A potential therapeutic target for the treatment of osteoarthritis and rheumatoid arthritis. | 4 |
| 38047304 | 2024 | TRPV1 enhances cholecystokinin signaling in primary vagal afferent neurons and mediates the central effects on spontaneous glutamate release in the NTS. | 0 |
Citation
Massimo Nabissi ; Giorgio Santoni
TRPV1 (transient receptor potential cation channel, subfamily V, member 1)
Atlas Genet Cytogenet Oncol Haematol. 2010-11-01
Online version: http://atlasgeneticsoncology.org/gene/50368/trpv1
