Lung: Pleuropulmonary blastoma
2010-07-01 Y Albert Yeh , Morris C Edelman Affiliation1.North Shore University Hospital, Long Island Jewish Medical Center, Hofstra University School of Medicine, New York, USA
Summary
Note
Pleuropulmonary blastoma is a rare aggressive malignant tumor of infancy and early childhood. The tumor arises in the lung and pleura and is regarded as a pulmonary dysontogenetic or embryonic neoplasm. It is the pulmonary analog of other tumors of childhood including Wilms tumor, neuroblastoma, hepatoblastoma, pancreatoblastoma, and retinoblastoma.
Synonyms include embryonal rhabomyosarcoma arising in congenital bronchogenic cyst, rhabdomyosarcoma arising in congenital cystic adenomatoid malformation, pulmonary sarcoma arising in mesenchymal cystic hamartoma, pulmonary blastoma associated with cystic lung disease of childhood.
Synonyms include embryonal rhabomyosarcoma arising in congenital bronchogenic cyst, rhabdomyosarcoma arising in congenital cystic adenomatoid malformation, pulmonary sarcoma arising in mesenchymal cystic hamartoma, pulmonary blastoma associated with cystic lung disease of childhood.
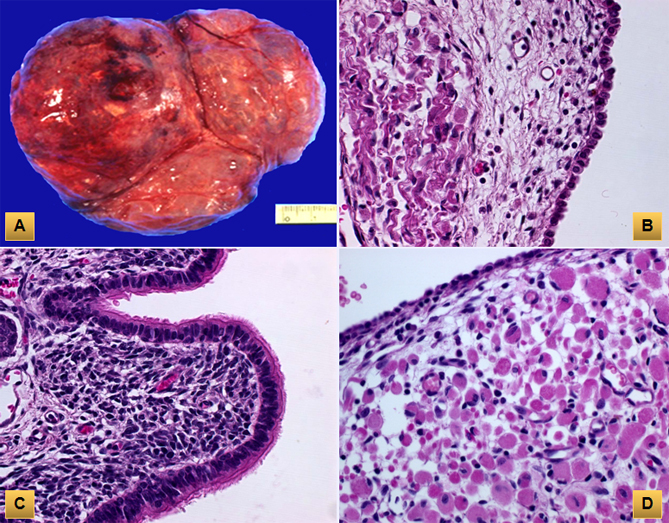
(A) Type I pleuropulmonary blastoma consists of multiloculated cyst filled with scanty clear serous fluid. (B) The cysts are lined by cuboidal epithelium resting on loose mesenchymal tissue. (C) There are areas of proliferating primitive cells with overlying stratified ciliated columnar epithelium. (D) Foci of rhabdomyoblasts with abundant eosinophilic cytoplasm and cross striations are noted.
Clinics and Pathology
Epidemiology
There are over 120 cases registered with The Pleuropulmonary Blastoma Registry (www.ppbregistry.org). The tumor affects mainly in children with age ranges from 1 month to 12 years. Most cases are diagnosed before 4 years of age. It can be found prenatally or present in older children and young adults. Males and females are equally affected.
Clinics
Approximately 25% of cases are associated with a heritable tumor syndrome. Additional tumors including cystic nephromas and other nephroblastic lesions, ovarian teratomas, multiple intestinal polyps, thyroid malignancies, medulloblastomas, and second pleuropulmonary blastomas have been observed in affected children. Clinical manifestations vary and depend on age and pathologic type of tumors. Most patients are presented with respiratory symptoms including dyspnea, chest pain, cough, and respiratory distress with or without pneumothorax. Asymptomatic lesions may be detected incidentally during investigation of unrelated clinical problems.
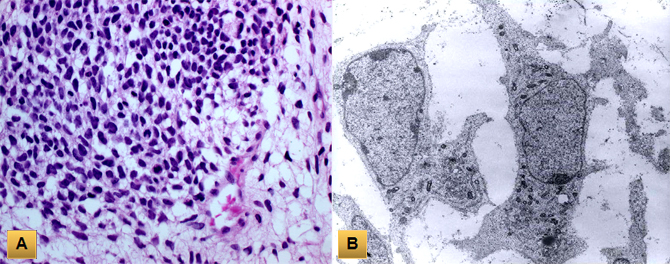
(A) Pleuropulmonary blastoma is composed of compact zones with primitive blastematous cells embedded in a loose myxoid stroma. (B) Electron microscopy shows spindle primitive cells with high nuclear-to-cytoplasmic ratio and oval nuclei.
Pathology
There are 3 pathologic types of pleuropulmonary blastoma with distinct macroscopic and microscopic features.
Type I pleuropulmonary blastoma is characterized by peripherally located, multicystic, and thin-walled structure. No solid nodules are observed within the thin-walled septae and multicystic spaces. Neoplastic cells can be identified beneath a benign cyst lining surface epithelium. This cambium layer-like zone contains proliferating primitive cells with mixed small, rounded, or spindled hyperchromatic nuclei. Rhabdomyoblasts are seen occasionally. Some tumors have immature cartilage within their fibrous septae.
Type II pleuropulmonary blastoma is a mixed solid and cystic tumor characterized by varied amount of thickened or nodule-like areas composed of rhabdomyosarcomatous or blastematous components.
Type III pleuropulmonary blastoma is characterized by a well-circumscribed, mucoid, white-tan solid mass attached to the pleura and involves a lobe or entire lung. Necrosis and hemorrhage are sometimes present in the friable areas. Histopathologically, type III tumor is an apparently heterogeneous tumor composed of one or more of the following elements: primitive blastema-like small cells with hyperchromatic nuclei with high nuclear-to-cytoplasmic ratio and abundant mitoses; spindled and ovoid cells embedded in a myxoid stroma; spindle cell sarcoma; nodules of immature or malignant chondroid elements. Isolated or clusters of large anaplastic cells with pleomorphic nuclei, atypical mitotic figures, or eosinophilic hyaline bodies are present.
Immunohistochemically, the neoplastic cells are stained positive for vimentin. CD117 (c-kit) and alpha-1-antitrypsin are focally positive in tumor cells. CD99 is weakly positive. Other immunostains including EMA, myogenin, S100, GFAP, neuron specific enolase, TTF-1, alpha-fetoprotein, chromogranin, and synaptophysin are negative. The pneumocytes lining the cysts and small airspaces are highlighted by cytokeratin. Muscle specific actin and desmin are expressed in rhabdomyoblasts and primitive cells in the subepithelial regions of cystic lesions.
Type I pleuropulmonary blastoma is characterized by peripherally located, multicystic, and thin-walled structure. No solid nodules are observed within the thin-walled septae and multicystic spaces. Neoplastic cells can be identified beneath a benign cyst lining surface epithelium. This cambium layer-like zone contains proliferating primitive cells with mixed small, rounded, or spindled hyperchromatic nuclei. Rhabdomyoblasts are seen occasionally. Some tumors have immature cartilage within their fibrous septae.
Type II pleuropulmonary blastoma is a mixed solid and cystic tumor characterized by varied amount of thickened or nodule-like areas composed of rhabdomyosarcomatous or blastematous components.
Type III pleuropulmonary blastoma is characterized by a well-circumscribed, mucoid, white-tan solid mass attached to the pleura and involves a lobe or entire lung. Necrosis and hemorrhage are sometimes present in the friable areas. Histopathologically, type III tumor is an apparently heterogeneous tumor composed of one or more of the following elements: primitive blastema-like small cells with hyperchromatic nuclei with high nuclear-to-cytoplasmic ratio and abundant mitoses; spindled and ovoid cells embedded in a myxoid stroma; spindle cell sarcoma; nodules of immature or malignant chondroid elements. Isolated or clusters of large anaplastic cells with pleomorphic nuclei, atypical mitotic figures, or eosinophilic hyaline bodies are present.
Immunohistochemically, the neoplastic cells are stained positive for vimentin. CD117 (c-kit) and alpha-1-antitrypsin are focally positive in tumor cells. CD99 is weakly positive. Other immunostains including EMA, myogenin, S100, GFAP, neuron specific enolase, TTF-1, alpha-fetoprotein, chromogranin, and synaptophysin are negative. The pneumocytes lining the cysts and small airspaces are highlighted by cytokeratin. Muscle specific actin and desmin are expressed in rhabdomyoblasts and primitive cells in the subepithelial regions of cystic lesions.
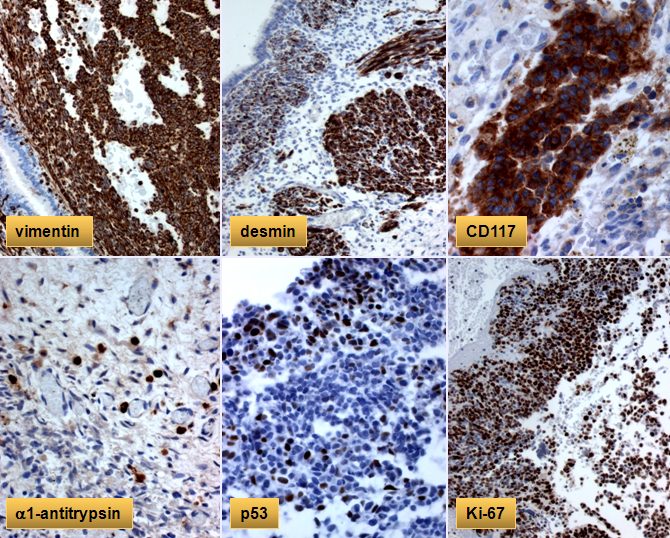
Immunohistochemical stains show strong positivity with vimentin, desmin, CD117, p53, and focal positivity with alpha-1-antitrypsin in the primitive tumor cells. The Ki-67 reveals high proliferative index of approximately 90%.
Cytogenetics
Gains of chromosome 8 detected by karyotyping and fluorescence in situ hybridization is the most consistent recurrent chromosomal aberration. Trisomy 2, unbalanced translocation between chromosomes 1 and X, and p53 mutations/deletions have been reported. One case with brain metastasis reveals an abnormal karyotype of 44,XY,del(1)(p12),-14der(19)add(19)(q13.3),-20. Heterozygous germline mutations in DICER1 are found in pleuropulmonary blastoma families.
Treatment
The current treatment for type I pleuropulmonary blastoma is complete surgical excision followed by combination chemotherapy with vincristine, actinomycin-D, and cyclophosphamide. Complete excision and adjuvant multi-agent chemotherapy are also applied for type II and type III tumors. Tumor progression from type I to type III has been documented.
Prognosis
Type I pleuropulmonary blastoma has the most favorable prognosis with a 5-year survival rate of 83%, whereas type II and III have a worse outcome of less than 50%.
Article Bibliography
| Pubmed ID | Last Year | Title | Authors |
|---|---|---|---|
| 7809508 | 1994 | Pleuropulmonary blastoma is THE pulmonary blastoma of childhood. | Dehner LP et al |
| 19556464 | 2009 | DICER1 mutations in familial pleuropulmonary blastoma. | Hill DA et al |
| 10357407 | 1999 | Pleuropulmonary blastoma in an adult: an initial case report. | Hill DA et al |
| 7277149 | 1981 | Embryonal rhabdomyosarcoma arising within a congenital bronchogenic cyst in a child. | Krous HF et al |
| 3048630 | 1988 | Pleuropulmonary blastoma. The so-called pulmonary blastoma of childhood. | Manivel JC et al |
| 17923214 | 2007 | Syndromic presentation of a pleuropulmonary blastoma associated with congenital cystic adenomatoid malformation. A case report. | Nur S et al |
| 9210721 | 1997 | Pleuropulmonary blastoma: a clinicopathologic study of 50 cases. | Priest JR et al |
| 11910518 | 2002 | Gains of chromosome 8 in pleuropulmonary blastomas of childhood. | Sebire NJ et al |
| 11779045 | 2001 | Gains of chromosome 8 are confined to mesenchymal components in pleuropulmonary blastoma. | Vargas SO et al |
| 9236843 | 1997 | Pleuropulmonary blastoma: fluorescence in situ hybridization analysis indicating trisomy 2. | Yang P et al |
Citation
Y Albert Yeh ; Morris C Edelman
Lung: Pleuropulmonary blastoma
Atlas Genet Cytogenet Oncol Haematol. 2010-07-01
Online version: http://atlasgeneticsoncology.org/solid-tumor/6040/lung-pleuropulmonary-blastoma
